Research Article - (2021) Volume 12, Issue 5
Background: This is a prospective, randomized, dose-controlled, double-blind, placebo-controlled phase II clinical study for the treatment of patients with painful diabetic peripheral neuropathy (pDPN) of the feet. Safety of cannabidiol (CBD) sublingual tablets was the primary objective. Efficacy of the proprietary water-solubilized CBD sublingual tablets on pain, sleep quality and anxiety were the secondary objectives. This trial was conducted remotely with a 28-day dosing regimen.
Methods: Subjects who met all inclusion/exclusion criteria and a Numerical Pain Rating Scale (NPRS) score of 5 or greater were recruited to participate. Trial subjects were randomized to receive either a 20 mg CBD sublingual tablet or placebo tablet three times a day (TID) for 28 days. Subjects were instructed to answer daily and weekly questions on the provided smart phone application.
Results: Subjects taking the CBD tablet reported statistically and clinically significant pain reduction from baseline as compared with placebo. They also had significantly more favorable Patient Global Impression of Change (PGIC) scores than the placebo group. Subjects in the CBD group met the Minimal Clinically Important Difference (MCID) for sleep improvement as compared with the placebo group. Clinically significant reduction in anxiety was also reported by subjects in the CBD group.
Conclusions: The results demonstrate that a proprietary formulation of water-solubilized 20 mg CBD sublingual tablets taken TID can achieve significant pain relief in chronic diabetics with pDPN, along with an improvement in sleep and anxiety reduction, without any tablet related adverse effects.
This study is listed on ClinicalTrials.Gov, ID: NCT04679545
Cannabidiol, CBD, Diabetes, Diabetic Peripheral Neuropathy, DPN, Sublingual Tablet, Pain, Placebo
Diabetic peripheral neuropathy (DPN) affects upwards of 50% of patients with diabetes [1]. According to the Centers for Disease Control and Prevention (CDC), 34.2 million people in the United States (US) have diabetes and another 88 million people have prediabetes [2]. Diabetes and its complications is the 7th leading cause of death and disability in the US [3].
DPN is damage to peripheral nerves in the stocking-glove distribution (feet and hands) due to chronic elevated blood sugar. The damage to the nerves results in debilitating pain that adversely impacts both quality of life and healthcare costs. According to the Journal of Diabetes Complications, the costs of painful DPN (pDPN) were approximately $30,000 per patient per year in 2015, far outpacing healthcare resource utilization and costs from patients with diabetes without pDPN [4]. Due to limited preventive options available to patients, safe and effective symptomatic management of pDPN is not only necessary but paramount.
The United States Food and Drug Administration (FDA) approved pharmaceutical treatments for pDPN are pregabalin, duloxetine, and tapentadol hydrochloride [5]. These medications have many severe adverse effects including a black box warning for suicidal ideation [6]. Other pharmaceutical classes of drugs used to treat pDPN are all off-label and include acetaminophen, non-steroidal anti-inflammatories, tricyclic anti-depressants, anti-seizure medications, opiate analgesics, and anti-arrhythmic medications [7]. Patients using these medications also experience serious sideeffects and with unreliable efficacy for symptom relief [8-14]. Physical modalities are also used to help mitigate the pain from DPN but again with inconsistent results [15].
There is limited clinical trial literature investigating the use of cannabis or cannabinoids for the treatment of pDPN. Pure Green Pharmaceuticals published a proof-of-concept, open label trial demonstrating that a proprietary water-solubilized 20 mg CBD sublingual tablet TID over a 21-day period could safely and effectively reduce the pain from DPN with no reported adverse effects [16]. That trial enrolled 31 subjects with pDPN; all subjects were required to have a Numerical Pain Rating Scale (NPRS) score of 5 or greater (moderate to severe chronic pain). Twenty-three of the 31 subjects were taking pain relieving medications specific for pDPN and continued them throughout the trial. A within-subjects analysis found that there was no significant difference between average NPRS scores in subjects on pain-relieving medications as compared with those who were not taking any pain-relieving medications (p<0.05). All subjects experienced a clinical and statistically significant improvement in their pain within 21 days (p<0.0001). In addition, the results of the study revealed clinically and statistically significant improvement in sleep quality and anxiety reduction across all subjects. The results of that trial helped to inform the next study examining whether a proprietary watersoluble sublingual CBD tablet may be used as a safe and effective treatment for pDPN versus a placebo [16].
The purpose of this trial was to determine whether a placebocontrolled design using the proprietary 20 mg CBD sublingual tablets TID would yield similar results as the aforementioned open-label trial. For this study, subjects were not permitted to take any other pain medicines beyond supplied study medication. The scope of the trial was limited to pDPN of the feet in order to focus the comparison of the test product to placebo control. This study was approved by an independent ethics committee and was carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). Written informed consent was obtained from all subjects prior to inclusion into the trial.
Fifty-five subjects diagnosed with pDPN of the feet were recruited to a 28-day randomized, double-blind, placebo-controlled, decentralized clinical trial in the US. After screening, eligible subjects were randomly assigned at a 1:1 ratio to receive a proprietary water-solubilized 20 mg CBD sublingual tablet or placebo sublingual tablet TID, for 28 days, where participation was conducted remotely at the subjects’ home.The inclusion and exclusion criteria for the study are available below. The exclusion criteria were updated to enhance the ability to recruit, although no one was recruited with the updated criteria.
The test product is a proprietary water-soluble sublingual 20 mg CBD tablet. The primary objective of the study was to evaluate safety to treat patients with pDPN of the feet, compared to a placebo control. The secondary objectives were to evaluate efficacy on pain, anxiety, and sleep quality compared to a placebo control, as well as to evaluate the subjects’ impression of their treatment response.
Rescue medication (acetaminophen or ibuprofen, per subject request) was provided to all participants, ad libitum when they desired additional pain relief.
Upon providing written informed consent to participate, subjects were immediately enrolled or were engaged in either a 7 or 14-day washout period prior to treatment if they were taking pain relief medication. The duration of the washout period was determined by the subject’s current pain medication use. At the conclusion of the washout period, subjects were asked to download a smart phone application to complete baseline measures, including a Pure Green Pharmaceuticals pain questionnaire utilizing a NPRS, the Pittsburgh Sleep Quality Index (PSQI), and the Zung Anxiety Self- Assessment Scale (SAS). Baseline medical history including clinical and demographic historical information were also collected. The NPRS options ranged from 0 (no pain) to 10 (worst possible pain) in order to assess the level of pain experienced by the subjects over the 28-day treatment period. Subjects were assessed for overall pain reported throughout the trial, as well as highest reportable pain at any given time during the study. The PSQI was scored out of a total of 21, where a higher score represents poorer sleep quality. The SAS raw score totals ranged from a possible 20 to 80 points, where a higher score represents higher anxiety levels. Raw score totals (20 – 80) were converted to an anxiety index (25 – 100) following completion of the assessment.
Each subject received a 28-day supply of their assigned sublingual tablet intervention (20 mg water-solubilized CBD or placebo) to be taken 3 times a day, 6 hours apart (morning, afternoon, and evening). Each subject recorded their pain score after taking a tablet and was also asked to complete a weekly NPRS assessment as they progressed throughout the trial. Additionally, subjects documented any changes to their concomitant medications, adverse events, and rescue medication usage throughout the trial. Following the 28-day treatment period, subjects were asked to recomplete the Pure Green Pharmaceuticals pain questionnaire, PSQI, and the SAS to compare post-treatment data with baseline information. In addition, subjects were asked to complete the Patient’s Global Impression of Change (PGIC) to evaluate their impression of their response to treatment. A final review of study drug tolerance and adverse events over the course of the treatment period was conducted.
Block randomization sequence was created using a computergenerated list using random block sizes of 4 and 6 with a 1:1 allocation of participants to treatment groups. The randomization sequence list was prepared by an employee with no clinical involvement in the trial and was concealed from the researcher enrolling and assessing participants for clinical trial entry. The sequence list was placed inside an opaque envelope and was kept in a locked storage container that was inaccessible to both the investigator and researcher. After the blinded researcher obtained the subject’s consent, the employee was contacted and was provided the subjects information for allocation consignment
Data were analysed using R Studio (Boston, MA). Statisticians were blinded during analysis and interpretation of results; the randomization code was broken after analysis and interpretation of results was completed. All data were summarized using descriptive statistics expressed as a mean (standard deviation (SD)) or median (interquartile range (IQR)) as appropriate for continuous variables, and number (percent) for categorical variables. A safety statistical endpoint assessment could not be conducted due to a complete lack of adverse events in the treatment group; there was one treatment emergent adverse event (TEAE) in the placebo group.
Change from baseline scores were calculated for pain, anxiety, and sleep. Prior to conducting the statistical analyses, each secondary endpoint was first assessed for normality. If the data were normally distributed, mean differences (MD) between treatment groups were evaluated using an independent samples t-test. If the data were not normally distributed, a Mann-Whitney U test was conducted to compare differences between treatment groups. Categorical variables were analysed using a chi-square test and the relative risk between treatment groups was calculated. All main analyses were conducted based on an intention to treat protocol that included all 54 randomized participants with baseline data. Missing follow-up data were imputed using the Multivariate Imputation by Chained Equations (MICE) R package. Ad hoc per-protocol sensitivity analyses were conducted with the three withdrawn participants removed for comparison. A p-value<0.05 was considered statistically significant.
Inclusion Criteria
Subjects must meet all of the following inclusion criteria to participate in this study:
1. Subject is at least 21 years of age;
2. Subject has a diagnosis of diabetic neuropathic pain of the feet determined by the subject’s primary care physician or related health care provider.
3. Subject has a mean pain scale score of ≥ 5 recorded in the 7 days prior to randomization.
4. If female, the subject is postmenopausal (>1 year), surgically sterile (>3 months), had a hysterectomy, or is currently using 2 effective forms of birth control.
5. Subject has not taken marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/ or CBD for at least 14 days prior to this study, and agrees to not take marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/or CBD while participating in this study.
6. If subject is currently taking gabapentin, pregabalin, or duloxetine, subject must be willing to and completes a 7-day washout of these medications prior to randomization.
7. Subject has not taken any NSAIDs and/or acetaminophen for at least 2 days prior to randomization.
8. Subject is willing to provide his/her written informed consent to participate in the study as stated in the informed consent document.
9. Subject is willing to use an electronic diary to enter trial information for 29 days.
Exclusion Criteria
Any subject who meets any of the exclusion criteria will be excluded from participation in this study:
1. Subject is pregnant or lactating;
2. Subject has an allergy to cannabis, the Cannabaceae plant family (e.g., hemp, hops), palmitoylethanolamide, or terpenes;
3. Subject has a known allergy to active or inert ingredients of the investigational product;
4. Subject is taking a concomitant medication or treatment that would complicate use or interpretation of the study drug’s effects (examples include: Cannabis or any cannabinoid products; Any drug or herbal product that influences the endocannabinoid system (ECS));
5. Subject is taking marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/ or CBD for at least 14 days prior to this study, and does not promise that they will not take marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/or CBD while participating in this study;
6. Subject currently resides in the state of Nebraska, Idaho, Iowa, or South Dakota.
7. Subject is currently being treated with antibiotics for sinus, throat, or lung infections;
8. Subject has shortness of breath associated with allergies; 9. Subject has uncontrolled asthma;
10. Subject has a fever and/or productive cough;
11. Subject has unstable angina, uncontrolled hypertension;
12. Subject currently or has a history of congestive heart failure;
13. Subject has any other unstable medical condition;
14. Subject has a personal or family history of schizophrenia;
15. Subject has a personal history or currently has suicidal ideation or attempted suicide;
16. Subject has a major neurological disorder, such as dementia, Parkinson’s disease, cognitive impairment, epilepsy, history of traumatic brain injury/head injury, and seizures.
17. Subject has taken pharmaceutical pain medicine of any kind, or has taken a NSAID and/or acetaminophen, within 2 days of randomization.
18. Subject has taken gabapentin, pregabalin, or duloxetine within 7 days prior to randomization or is unwilling to stop these medications.
19. Subject has an allergy to, or has an intolerance to, NSAIDs or acetaminophen.
20. Subject is currently taking any form of opioids.
21. Subject has a history of substance or alcohol abuse.
22. Subject has clinically significant illness, including cardiovascular disorders.
23. Subject has any condition in which the investigator believes will confound the data of the study or could put the subject at risk of harm.
24. Subject does not have access to a smart phone or does not know how to use a smart phone application.
Results
Screening: A total of 226 patients were screened between January 6 and March 3, 2021, 171 of whom were ineligible (Table 1). Of these patients, 170 were determined to be ineligible prior to entering the screening period and one withdrew consent during the washout period prior to randomization. There were 55 participants randomized, of whom one withdrew consent prior to completing the baseline assessment and was therefore not included in analysis. The 54 participants included in analysis were randomized equally between the active and placebo groups (Figure 1).
| Exclusion Criteria | N=171 n (%) |
|---|---|
| Unwilling to complete 14-day cannabis washout* | 43 (25.1%) |
| Unwilling to complete 7-day medication washout | 34 (19.9%) |
| Unwilling to consent | 33 (19.3%) |
| Taking any form of opioids | 15 (8.8%) |
| Pain <5 | 14 (8.2%) |
| Clinically significant illness, including cardiovascular disorders | 5 (2.9%) |
| Personal history of suicidal ideation or suicide attempts | 4 (2.3%) |
| Taking any anti-anxiety or anti-depression medications | 4 (2.3%) |
| Other | 3 (1.8%) |
| History of substance or alcohol abuse | 3 (1.8%) |
| Shortness of breath associated with allergies | 3 (1.8%) |
| Major neurological disorder | 2 (1.2%) |
| No DPN of the feet | 1 (0.6%) |
| Currently pregnant or lactating | 1 (0.6%) |
| Cannabis allergy | 1 (0.6%) |
| Lives in Nebraska, Idaho, Iowa or South Dakota | 1 (0.6%) |
| Uncontrolled asthma | 1 (0.6%) |
| Other unstable medical condition | 1 (0.6%) |
| Personal or family history of schizophrenia | 1 (0.6%) |
| Allergy to, or an intolerance to, NSAIDs or acetaminophen | 1 (0.6%) |
| *One patient entered the washout period and withdrew as they were unwilling to complete the cannabis washout. | |
Table1: Reasons for ineligibility.
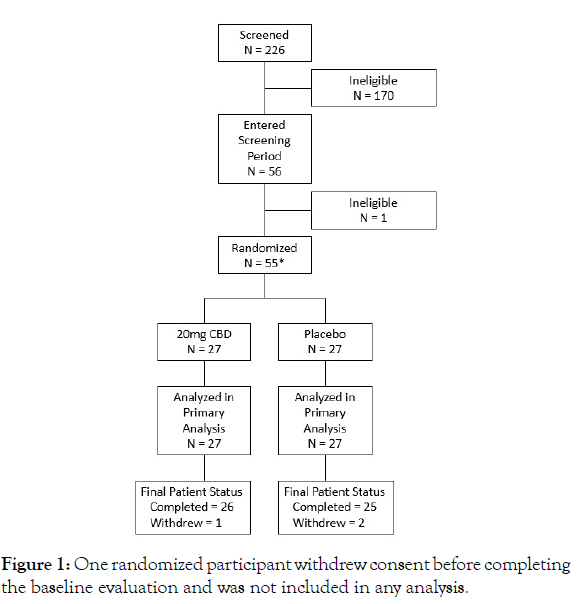
Figure 1: One randomized participant withdrew consent before completing the baseline evaluation and was not included in any analysis.
Demographics: The average age of participants was 52.62 years (SD=10.79), and 59.3% were women (Table 2). The majority of participants were either white (55.6%) or Black (38.9%), and very few identified as Hispanic (5.6%). The average BMI was 31.97, classified as obese, and almost all participants reported a comorbid health condition (96.3%). The average time since participants’ DPN diagnosis was 7.56 years (SD = 6.62) with the most common descriptions of pain being sharp/stabbing (90.7%) and tingling (90.7%).
| Characteristic | Total (N= 54) Mean (SD) or n (%) |
20 mg CBD Group (N = 27) Mean (SD) or n (%) |
Placebo Group (N = 27) Mean (SD) or n (%) |
|---|---|---|---|
| Age | 52.62 (10.79) | 52.85 (10.94) | 52.4 (10.8) |
| Gender Female Male |
32 (59.3%) 22 (40.7%) |
15 (55.6%) 12 (44.4%) |
17 (63.0%) 10 (37.0%) |
| BMI | 31.97 (7.70) | 30.55 (6.44) | 33.38 (8.67) |
| Race White / Caucasian Black / African American Other |
30 (55.6%) 21 (38.9%) 3 (5.5%) |
14 (51.9%) 11 (40.7%) 2 (7.4%) |
16 (59.3%) 10 (37.0%) 1 (3.7%) |
| Ethnicity Non-Hispanic Hispanic |
51 (94.4%) 3 (5.6%) |
26 (96.3%) 1 (3.7%) |
25 (92.6%) 2 (7.4%) |
| Marital Status Married Single Divorced Widowed Living as married Separated |
22 (40.7%) 15 (27.8%) 10 (18.5%) 4 (7.4%) 2 (3.7%) 1 (1.9%) |
8 (29.7%) 9 (33.3%) 6 (22.2%) 2 (7.4%) 1 (3.7%) 1 (3.7%) |
14 (51.9%) 6 (22.2%) 4 (14.8%) 2 (7.4%) 1 (3.7%) 0 (0%) |
| Highest level of education Some college College degree High school Graduate/professional degree |
24 (44.4%) 16 (29.6%) 9 (16.7%) 5 (9.3%) |
11 (40.8%) 9 (33.3%) 3 (11.1%) 4 (14.8%) |
13 (48.1%) 7 (26.0%) 6 (22.2%) 1 (3.7%) |
| Occupation status Active Retired |
32 (59.3%) 22 (40.7%) |
15 (55.6%) 12 (44.4%) |
17 (63.0%) 10 (37.0%) |
| Comorbid conditions Yes No |
52 (96.3%) 2 (3.7%) |
27 (100%) 0 (0%) |
25 (92.6%) 2 (7.4%) |
| Time since DPN diagnosis | 7.56 (6.62) | 7.22 (5.71) | 7.89 (7.50) |
| Description of pain in feet Sharp/stabbing Tingling Dull/achy Burning Cold Squeezing/pressure None of the above |
49 (90.7%) 49 (90.7%) 30 (55.6%) 30 (55.6%) 30 (55.6%) 19 (35.2%) 0 (0%) |
24 (88.9%) 26 (96.3%) 15 (55.6%) 16 (59.3%) 14 (51.9%) 6 (22.2%) 0 (0%) |
25 (92.6%) 23 (85.2%) 15 (55.6%) 14 (51.9%) 16 (59.3%) 13 (48.1%) 0 (0%) |
| NPRS (0-10) Average pain Highest pain |
7.31 (1.18) 8.72 (1.09) |
7.19 (1.18) 8.74 (1.16) |
7.44 (1.19) 8.70 (1.03) |
| PSQI (0-21) | 10.72 (3.71) | 11.1 (3.43) | 10.3 (3.99) |
| SAS (20-80) Anxiety index (25-100) |
38.98 (8.53) 48.91 (10.69) |
38.5 (8.50) 48.3 (10.60) |
39.4 (8.69) 49.5 (10.90) |
| SD, Standard deviation; BMI, body mass index; DPN, diabetic peripheral neuropathy; NPRS, Numeric Pain Rating Scale; PSQI, Pittsburgh Sleep Quality Index; SAS, Anxiety Self-Assessment Scale. | |||
Table 2: Baseline Characteristics of Trial Subjects.
Compliance & rescue medication use: Data regarding the proportion of participants who were at least 80% compliant with taking their randomized tablets and the use of rescue medication are summarized in Table 3. There was no statistically significant difference between groups in the proportion of compliant participants (96.3% vs. 92.6% in the 20 mg CBD and placebo group, respectively). In terms of the use of rescue medication at any point during the study, there was no significant difference between treatment groups (48.1% vs. 51.9% in the 20 mg CBD and placebo group, respectively). Of note, in both groups, the proportion of participants who required rescue medication decreased over time and, though not statistically significant, the proportion was lower in the 20 mg CBD group at each timepoint. There was also no significant difference in the daily amount of rescue medication consumed across the whole group (Table 3), or among only the 26 participants who required rescue medication (Table 4). The perprotocol population demonstrated similar findings (Table 5).
| Total (N= 54) Median (IQR) or n (%) |
20 mg CBD Group (N = 27) Median (IQR) or n (%) |
Placebo Group (N = 27) Median (IQR) or n (%) |
20 mg CBD vs. Placebo | |
|---|---|---|---|---|
| At least 80% compliant | 51 (94.4%) | 26 (96.3%) | 25 (92.6%) | RR: 1.04 [0.91, 1.18]; p = 1.00* |
| Use of rescue medication Any 1-7 days 8-14 days 15-21 days 22-28 days |
27 (50.0%) 24 (44.4%) 21 (38.9%) 16 (29.6%) 9 (16.7%) |
13 (48.1%) 11 (40.7%) 9 (33.3%) 6 (22.2%) 2 (7.4%) |
14 (51.9%) 13 (48.1%) 12 (44.4%) 10 (37.0%) 7 (25.9%) |
RR: 0.93 [0.54, 1.58]; p = 0.79 RR: 0.85 [0.46, 1.54]; p = 0.58 RR: 0.75 [0.38, 1.48]; p = 0.40 RR: 0.60 [0.25, 1.42]; p = 0.23 RR: 0.29 [0.07, 1.25]; p = 0.14* |
| Median daily amount of rescue medication (milligrams) | 7.1 (0.0, 138.4) |
0.0 (0.0, 98.2) |
14.3 (0.0, 134.0) |
W = 341.5; p = 0.68 |
| IQR, interquartile range; RR, relative risk; W, Wilcoxon W statistic. [] = 95% confidence interval *Based on Fisher's exact test as expected cell count was <5. |
||||
Table 3: Treatment Compliance and Use of Rescue Medications of Trial Subjects.
| Total (N=26) Mean ± SD |
20 mg CBD Group (N=12) Mean ± SD |
Placebo Group (N=14) Mean ± SD |
20 mg CBD vs. Placebo | |
|---|---|---|---|---|
| Mean daily amount of rescue medication (milligrams) | 150.0 ± 99.1 | 147.0 ± 109.0 | 153.0 ± 94.3 | MD: -6.00 [77.86, -88.92]; p = 0.89 |
| MD, mean difference; SD, standard deviation. []=95% confidence interval |
||||
Table 4: Post-hoc analysis of daily amount of rescue medication among participants who required rescue medication in intention to treat analysis.
| Total (N=51) Median (IQR) or N (%) |
20 mg CBD Group (N=26) Median (IQR) or N (%) |
Placebo Group (N=25) Median (IQR) or N (%) |
20 mg CBD vs. Placebo | |
|---|---|---|---|---|
| Use of rescue medication Any 1-7 days 8-14 days 15-21 days 22-28 days |
26 (51.0%) 23 (45.1%) 21 (41.2%) 16 (31.4%) 9 (17.6%) |
12 (46.2%) 10 (38.5%) 9 (34.6%) 6 (23.1%) 2 (7.7%) |
14 (56.0%) 13 (52.0%) 12 (48.0%) 10 (40.0%) 7 (28.0%) |
RR: 0.82 [0.48, 1.42]; p = 0.48 RR: 0.74 [0.40, 1.37]; p = 0.33 RR: 0.72 [0.37, 1.41]; p = 0.33 RR: 0.58 [0.25, 1.35]; p = 0.19 RR: 0.27 [0.06, 1.20]; p = 0.08* |
| Median daily amount of rescue medication (milligrams) | 14.3 (0.0, 142.9) |
0.0 (0.0, 121) |
50.0 (0.0, 143.0) |
W = 290; p = 0.49 |
| IQR, interquartile range; RR, relative risk; W, Wilcoxon W statistic. []=95% confidence interval *Based on Fisher's exact test as expected cell count was <5. |
||||
Table 5: Use of treatment intervention for 51 participants included in the per protocol analysis.
Primary endpoint
Treatment emergent adverse events: There was only one TEAE reported during the trial; a participant in the placebo group reported gastrointestinal upset of moderate severity. At the time of the report the investigators determined it was not serious and not related to the intervention; however, the participant subsequently withdrew consent and ended their participation in the trial.
Secondary endpoints: Data regarding pain, sleep, anxiety, and PGIC outcomes are summarized in Table 6 with baseline values reported in Table 2. The average pain score for the sample at baseline was 7.31 (SD=1.18), with the highest pain reported at 8.72 (SD=1.09). Participants in the 20 mg CBD group had significantly greater pain reductions at final follow-up on both the NPRS for average pain (-3.56 vs. -1.74 in the 20 mg CBD and placebo group, respectively; p < 0.01; Figure 1) and NPRS for highest pain (-3.70 vs. -1.85 in Groups 1 and 2, respectively; p<0.01; Figure 2). The minimal clinically important difference (MCID) for pain that has been reported in the literature for individuals with painful DPN is 2 points on an NPRS [17], of which the 20 mg CBD group exceeded for both average pain and highest pain.
| Outcome | 20 mg CBD Group (N=27) Mean ± SD |
Placebo Group (N=27) Mean ± SD |
20 mg CBD vs. Placebo |
|---|---|---|---|
| NPRS for average pain (0-10) |
-3.56 ± 1.80 | -1.74 ± 1.40 | MD: -1.82 [-0.93, -2.70]; p = 0.0001171 |
| NPRS for highest pain (0-10) |
-3.70 ± 2.37 | -1.85 ± 1.66 | MD: -1.85 [-0.73, -2.97]; p = 0.0002856 |
| PSQI (0-21) | -3.41 ± 5.11 | -2.70 ± 3.91 | MD: -0.71 [1.78, -3.19]; p = 0.57 |
| SAS (20-80) Anxiety index (25-100) |
-4.37 ± 5.87 -5.26 ± 7.29 |
-3.11 ± 6.17 -4.22 ± 7.89 |
MD: -1.26 [2.03, -4.55]; p = 0.45 MD: -1.04 [3.11, -5.18]; p = 0.62 |
| PGIC Question 1 (1-7) PGIC Question 2 (0-10) |
4.70 ± 1.20 3.04 ± 1.34 |
3.70 ± 1.59 4.26 ± 1.81 |
MD: 1.00 [0.23, 1.77]; p = 0.01 MD: -1.22 [-0.35, -2.09]; p = 0.01 |
| MD, mean difference; SD, standard deviation; PGIC, Patient’s Global Impression of Change; NPRS, Numeric Pain Rating Scale; PSQI, Pittsburgh Sleep Quality Index; SAS, Zung Anxiety Self-Assessment Scale. []=95% confidence interval All values apart from the PGIC are change from baseline to 28 days. For all outcomes, except the PGIC, negative values represent a more favorable outcome. A higher score is more favorable for PGIC Question 1, and a lower score is more favorable for PGIC Question 2. |
|||
Table 6: Pain, Sleep, Anxiety, and Impression of Change Outcomes for Trial Subjects.
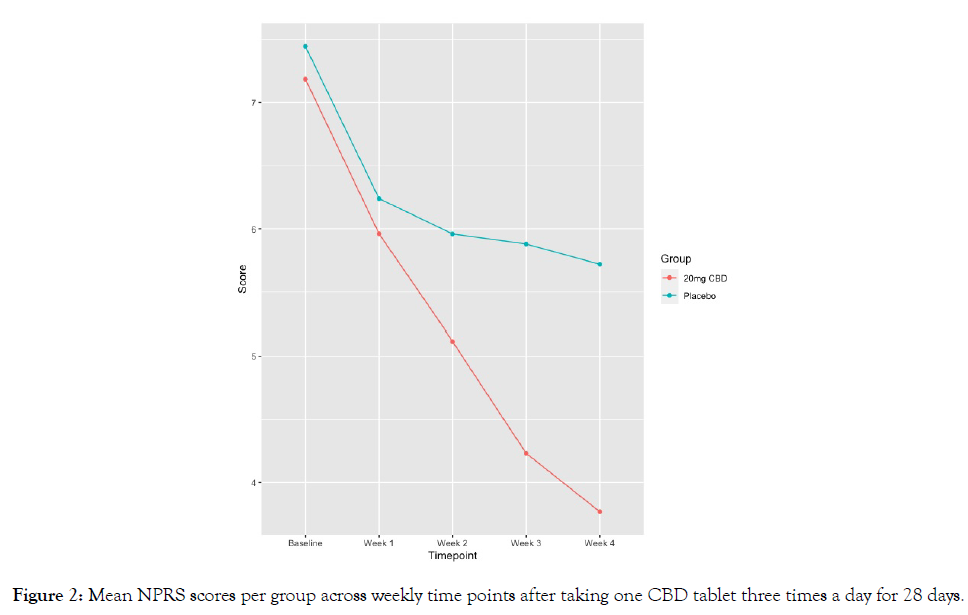
Figure 2: Mean NPRS scores per group across weekly time points after taking one CBD tablet three times a day for 28 days.
The average baseline sleep score of 10.72 (SD=3.71) as assessed by the PSQI corresponded to poor sleep quality, and although both groups reported an improvement in their score at followup, the difference was not statistically significant between them. However, the average change among the 20 mg CBD group, -3.41 (SD=5.11) was greater than the MCID of 3 for the PSQI that has been previously reported in the literature [18]. The average change in scores for the placebo group (-2.70, SD=3.91) did not meet the MCID.
The average baseline anxiety index score of 48.91 (SD=10.69) on the SAS was reflective of minimal to moderate anxiety. Similar to the PSQI, both groups reported an improvement in their score at follow up, but the difference was not statistically significant between them. Of note, the average anxiety index score for the 20 mg CBD group fell below the cutoff representing anxiety within normal range (<45) at follow-up, whereas the placebo group did not (Figure 3).
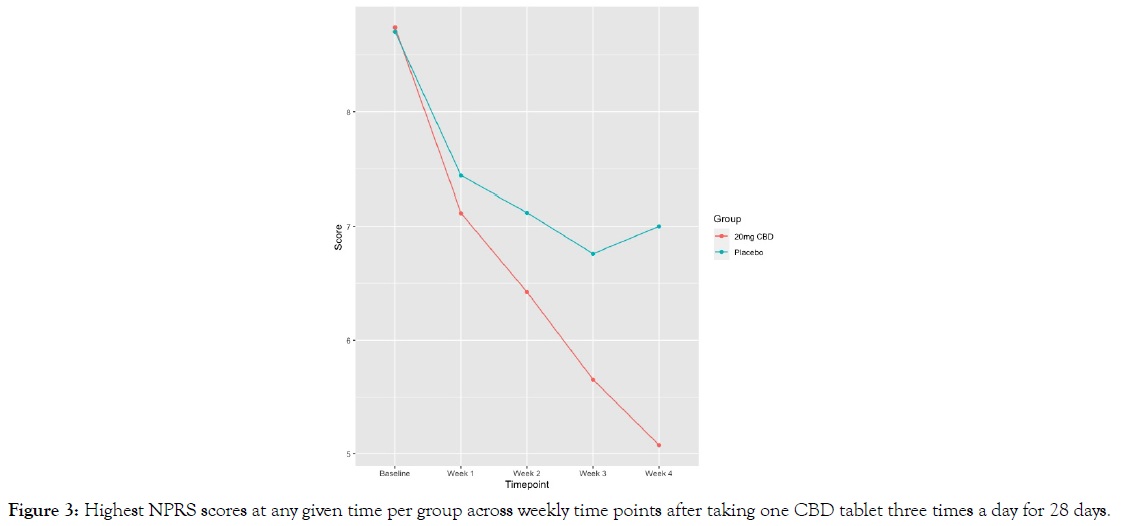
Figure 3: Highest NPRS scores at any given time per group across weekly time points after taking one CBD tablet three times a day for 28 days.
At the conclusion of the trial, patients’ impression of their response to treatment was measured using the PGIC questionnaire. Participants in the 20 mg CBD group exhibited statistically significant favorable PGIC scores as compared with the placebo (p=0.01). The per-protocol population analysis demonstrated similar findings for all endpoints (Table 7).
| Outcome | 20mg CBD Group (N=26) Mean ± SD |
Placebo Group (N=25) Mean ± SD |
20mg CBD vs. Placebo |
|---|---|---|---|
| NPRS for average pain (0-10) |
-3.54 ± 1.84 | -1.72 ± 1.46 | MD: -1.82 [-0.89, -2.75]; p = 0.0002829 |
| NPRS for highest pain (0-10) |
-3.81 ± 2.35 | -1.68 ± 1.57 | MD: -2.13 [-1.00, -3.25]; p = 0.000426 |
| PSQI (0-21) | -3.73 ± 4.92 | -2.84 ± 3.94 | MD: -0.89 [1.62, -3.40]; p = 0.48 |
| SAS (20-80) Anxiety index (25-100) |
-4.23 ± 5.94 -5.31 ± 7.43 |
-3.16 ± 6.42 -4.16 ± 8.08 |
MD: -1.07 [2.42, -4.56]; p = 0.54 MD: -1.15 [3.23, -5.52]; p = 0.60 |
| PGIC Question 1 (1-7) PGIC Question 2 (0-10) |
4.69 ± 1.23 2.96 ± 1.31 |
3.64 ± 1.63 4.36 ± 1.82 |
MD: 1.05 [0.24, 1.87]; p = 0.01 MD: -1.40 [-0.50, -2.30]; p = 0.00308 |
| MD, mean difference; SD, standard deviation; PGIC, Patient’s Global Impression of Change; NPRS, Numeric Pain Rating Scale; PSQI, Pittsburgh Sleep Quality Index; SAS, Anxiety Self-Assessment Scale [] = 95% confidence interval All values apart from the PGIC are change from baseline to 28 days. For all outcomes, except the PGIC, negative values represent a more favorable outcome. A higher score is more favorable for PGIC Question 1, and a lower score is more favorable for PGIC Question 2. |
|||
Table 7: Pain, sleep, anxiety, and impression of change outcomes for 51 participants included in the per protocol analysis.
The results of this trial met the primary safety endpoint of the 20 mg CBD group as compared to placebo. Over the course of the 28- day treatment period, there were no adverse effects reported in the CBD group. There was, however, one TEAE reported in the placebo group that was clearly unrelated to treatment. This highlights the safety of this proprietary water-soluble 20 mg sublingual CBD tablet when taken TID for a minimum of 28 days.
There was a statistically significant reduction in pain between the CBD group and the placebo group in both average pain and highest pain reported throughout the trial. Interestingly, the results of this placebo-controlled trial mirrored those of the Pure Green Pharmaceuticals open-label pDPN trial [16] where both studies revealed a significant drop in NPRS scores by approximately 50%. In addition, the MCID for NPRS was exceeded by the CBD group and not the placebo group, further illustrating the efficacy of the CBD tablet in reducing pain associated with pDPN of the feet.
Sleep outcomes revealed a clinical improvement in the CBD group as compared with the placebo group. The MCID for the PSQI was exceeded in the CBD group, suggesting a clinical effectiveness for the 20 mg CBD tablet for sleep.Although the placebo group reported a slight improvement in sleep quality, the improvement was insignificant and did not reach the minimum criteria for clinical efficacy. While the CBD group showed clinical efficacy over the placebo group for sleep improvement, there was no statistically significant difference in PSQI scores.
Similar to the sleep quality outcomes, the clinical interpretation of the SAS anxiety index scores favored the CBD group. Both groups pre-treatment scores were in the category of minimal to moderate anxiety. Post-treatment anxiety scores revealed that the CBD group changed to the category of within normal range, where as the placebo group did not change and remained in the category of minimal to moderate anxiety at the conclusion of the trial. Although posttreatment SAS scores showed a clinical effectiveness in the reduction of anxiety for the CBD group, the difference of scores were not statistically significant.
The statistically significant result in PGIC scores highlights the overall efficacy and success of the CBD tablet for pDPN. This led to a clinically significant improvement in quality of life that was not realized in the placebo group. It is important to note that medications not only need to be successful in efficacy, but also need to be well tolerated without adverse effects; the 20 mg CBD tablet achieved these goals. Anecdotally, subjects were eager to participate in this trial because they were displeased with the adverse effects and lack of efficacy experienced with their pDPN medications.
Considering the positive results of this trial, there were some inherent limitations to a randomized, placebo-controlled design. As noted in Table 1, approximately 20% of ineligible subjects chose not to participate as they were unwilling to discontinue their existing pDPN medications for the random chance of being placed in the placebo group. Additionally, the results of a randomized trial do not necessarily mimic real life treatment situations due to its highly controlled setting and limiting exclusion criteria. Therefore, additional research with broader entry criteria that reflects a more inclusive clinical population is needed.
This placebo-controlled, double-blind, randomized trial validated the results of Pure Green Pharmaceutical’s open label pDPN trial [16] as well as demonstrated the safety and efficacy of the water-solubilized 20 mg CBD sublingual tablet for the treatment of pDPN. This 28-day trial revealed statistically and clinically significant improvement in pain and a clinically significant improvement in sleep quality and in anxiety reduction for those in the CBD treatment group. Additionally, subjects taking CBD affirmed these results by having a statistically significant greater response to treatment as compared with subjects taking placebo. The benefit of this study demonstrates that the sublingual 20 mg CBD tablet should be considered as a safe and effective treatment for pDPN.
Design and analyses: D.K., M.C., S.G.; manuscript writing: D.K., M.C., S.G.; manuscript revising for intellectual content: S.G. All authors approved the final version of the manuscript.
The authors are associated with Pure Green Pharmaceuticals, Inc, which developed and manufactured the Pure Green® CBD 20 mg tablets.
This work was solely financed by Pure Green Pharmaceuticals, Inc.
The authors would like to thank all investigators, study teams, and patients for participating in this study
Citation: Kimless D, Caloura MK, Goldner S. (2021) Cannabidiol (CBD) For the Treatment of Painful Diabetic Peripheral Neuropathy of The Feet: A Placebo-Controlled, Double-Blind, Randomized Trial. J Diabetes Metab. 12:870.
Received: 17-Mar-2021 Published: 31-May-2021, DOI: 10.35248/2155-6156.21.12.879
Copyright: © 2021 Kimless D, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.