Review Article - (2021) Volume 9, Issue 7
This paper determines the study of dosing and treatment therapy and drug effectiveness used for hospitalized COVID-19 patients whom are being administered Remdesivir in a large, multi-center, clinical trial led by NIH’s National Institute of Allergy and Infectious Diseases (NIAID). Remdesivir has been as of late a solid contender for the treatment of COVID-19 and in this critique, some of scientist depicted the structure of the coronavirus in a straightforward manner just as the site where Remdesivir acts whereas in continuous clinical preliminaries, just as a distributed report that was led on merciful base and The COVID-19 pandemic may wean somewhere near the finish of summer 2020, yet the danger of regularity exists. Hence, future removal of operators, for example, Remdesivir may be critical for guaranteeing a proficient treatment, decline mortality and permit early release.
Remdesivir is commonly known as GS-5734 ,While the principal medication cleared for the treatment of COVID-19, chiefly profited more advantageous patients who weren't reliant on ventilators or heart-lung sidestep machines, as per distributed aftereffects of the examination used to get the medication available and the medication helped patients tainted with the novel coronavirus recuperate quicker, permitting them to get back after around 11 days, contrasted with 15 days for the individuals who were treated with a fake treatment, as indicated by the report in the New England Journal of Medicine [1]. There were likewise signs the medication expanded their endurance rate-7.1% on patients on Remdesivir and 11.9% on a fake treatment passed on inside about fourteen days [2]. All things considered, the distinction wasn't measurably noteworthy, which means it could have originated from possibility while some outside specialists invited the discoveries, saying the outcomes indicated patients on Remdesivir cut their recuperation time by 27% where few speculators regretted the absence of data that was at first accessible [3]. The principal take a gander at the discoveries came when Anthony Fauci, the leader of the National Institute of Allergy and Infectious Diseases, gave a review of the discoveries during an occasion in the Oval Office with President Donald Trump a month ago. RDV attach to RdRp and behave as RNA chain terminator and which exhibits proper effect in vitro procedure against SARS-CoV-2 with an EC50 at 48 hours of 0.77 μM in Vero E6 cells, where some comparable activity has been shown against other zoonotic (diseases transfer from animal humans) COVID-19 with EC50 values of 0.07 μM demonstrated for both SARS-COV-1 and MERS-COVID [4].
Remdesivir clinical preliminary program was intended to address different logical inquiries in equal, to quickly assess the potential for Remdesivir to treat COVID-19 patients.
As there are several trails which are going on the Remdesivir.
• Gilead SIMPLE study in patients with severe disease
• Gilead SIMPLE study in patients with moderate disease
• NIAID study
• WHO Solidarity trial
• Inserm Discovery trial
Gilead-initiated “SIMPLE” trials
Gilead is leading two randomized, open-name, multicenter, Phase 3 clinical examinations to assess the security and adequacy of two dosing spans-5 days and 10 days of Remdesivir in grownups determined to have COVID-19 as well as there are several other trails performed in various country by various organizations-The U.S National Institute of Allergy and Infectious Diseases (NIAID), some portion of the National Institutes of Health, led a Phase 2 versatile, randomized, twofold visually impaired, fake treatment controlled preliminary assessing Remdesivir as a potential treatment for hospitalized grown-up patients determined to have COVID-19. Gilead gave study sedate at no charge and gave contribution on study plan and direct. The World Health Organization is directing a versatile, randomized, open-name, multi-focus clinical preliminary of the security and adequacy of Remdesivir and three other investigational medicines in hospitalized grown-ups determined to have COVID-19. Gilead is giving investigation medicate at no charge and gave contribution on study plan and lead. Furthermore, Inserm in France is leading a European report assessing Remdesivir and other potential medicines, utilizing an ace convention created by WHO. Gilead is giving investigation tranquilize at no charge and gave contribution on study structure and lead. Wellbeing experts in China led two clinical preliminaries in patients who had been contaminated with COVID-19 to decide the security and adequacy of Remdesivir as a potential treatment for the coronavirus [5]. These two investigations were halted ahead of schedule because of low enlistment. The two investigations were composed by the China-Japan Friendship Hospital and were being led at numerous locales in Hubei territory. Gilead gave study tranquilize at no charge and gave contribution on study plan and direct (Figure 1).
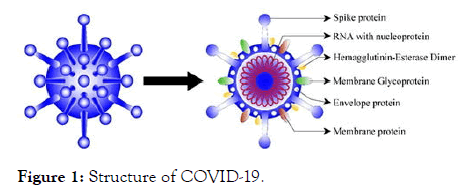
Figure 1: Structure of COVID-19.
To grow the flexibly of Remdesivir over the globe, Gilead Sciences has marked non-elite willful authorizing concurrences with four Indian conventional pharma producers, in particular; Cipla, Hetero Labs, Jubilant Lifesciences and Mylan just as a Pakistani firm, Ferozsons Laboratories to fabricate and disseminate Remdesivir in 127 nations. These nations comprise of almost all low-pay and lower-center pay nations, just as a few upper-center and high-pay nations that face critical obstructions to medicinal services get to. In any case, purportedly, there are four to five Bangladeshi firms that have just begun fabricating the counter popular medication Remdesivir. Organizations like Eskayef Pharmaceuticals and Beximco Pharmaceuticals have in actuality propelled their items for the COVID-19 treatment under brand names, Remivir and Bemsivir individually [6]. Be that as it may, in an email reaction, Gilead Sciences explained theories identified with the award of assembling licenses of its Remdesivir image to Bangladeshi firms and expressed, "Gilead has not given a permit to Beximco Pharma, Eskayef Pharmaceuticals or some other organization in Bangladesh to make Remdesivir. Gilead can't remark on or check the credibility or viability of this item as it isn't fabricated by Gilead or one of our authorized accomplices." Considering, the rising number of COVID-19 positive cases in the Maharashtra State, after (DCGI) endorsement for Remedisivir in India, the Maharashtra Government had made a declaration that it will acquire 10,000 vials of Remdesivir tranquilize from Eskayef Pharmaceuticals at a cost $160 per vial for example roughly Rs 12000. In any case, after the Gilead proclamation, the express government's choice to secure the medication from Eskayef Pharma has been required to be postponed [7,8].
Research by researchers at Götte's lab found that the utilization of polymerase protein extricated from coronavirus, MERS taking note of that the chemicals can fuse Remdesivir into the incorporating single-abandoned RNA Remdesivir looking like as a RNA building square turns into a piece of the new RNA strand. It was noticed that in the wake of including Remdesivir, the polymerase compound quits including more RNA subunits, halting genome replication. These instruments are connected to Remdesivir's capacity to use into a functioning structure known as GS-441524 which is an adenosine nucleotide analog.The GS-441524 meddles with the activity of viral RNA-subordinate RNA polymerase and avoids editing by viral Exoribonuclease (ExoN). This abatements viral RNA production studies are as yet continuous to comprehend whether the prodrug ends RNA chains or it causes changes in the RNA. Using Ebola infection considers, it was noticed that the prodrug, Remdesivir, hindered the activity of RNA-subordinate RNA polymerase, causing the prolongation of the blended chain [9].
Remdesivir is a prodrug that uses into its dynamic structure GS-441524. An adenosine nucleotide simple, GS-441524 meddles with the activity of viral RNA-subordinate RNA polymerase and dodges editing by viral Exoribonuclease (ExoN), causing a lessening in viral RNA production. Though in some infections, for example, the respiratory syncytial infection yet not Ebola infection, it causes the RNA-subordinate RNA polymerases to stop, its transcendent impact is to prompt an irreversible chain end. Not at all like with numerous other chain eliminators, this was not interceded by forestalling option of the quickly resulting nucleotide, yet is rather postponed, happening after five extra bases have been added to developing RNA chain. Changes in the mouse hepatitis infection RNA replicate that prompt incomplete protection from Remdesivir were recognized in 2018. These transformations make the infections less compelling in nature and the scientists accept they will probably not continue where the medication isn't being used.
Our preliminary found that intravenous Remdesivir didn't essentially improve the chance to clinical improvement, mortality, or time to freedom of infection in patients with genuine COVID-19 contrasted and fake treatment [10-15]. Contrasted and a past investigation of merciful utilization of Remdesivir, our examination populace was less sick (eg, at the hour of enrolment, 0.4% were on obtrusive mechanical ventilation or extracorporeal layer oxygenation versus 64% in the past investigation) and was dealt with to some degree prior in their malady course (middle 10 days versus 12 days) [16]. Such contrasts may be required to support Remdesivir, giving more prominent impacts in our examination populace, yet our outcomes didn't meet this desire. Be that as it may, our investigation didn't arrive at its objective enrolment in light of the fact that the tough general wellbeing estimates utilized in Wuhan prompted checked decreases in new patient introductions in mid-March, and limitations on medical clinic bed accessibility brought about most patients being enlisted later over the span of ailment. Therefore, we couldn't sufficiently evaluate whether prior Remdesivir treatment may have given clinical advantage. Be that as it may, among patients who were treated inside 10 days of side effect beginning, Remdesivir was not a critical factor but rather was related with a numerical decrease of 5 days in middle chance to clinical improvement. Progressing controlled clinical preliminaries are relied upon to affirm or invalidate our discoveries. In one murine model of SARS, Remdesivir treatment beginning at 2 days after contamination, after infection replication and lung aviation route epithelial harm had just topped, fundamentally diminished SARS-CoV-1 lung titres however didn't diminish illness seriousness or mortality. A requirement for early treatment has been found in non-human primate models of SARS and MERS in which infection replication is exceptionally brief and lung pathology seems to grow more quickly than in human infections. Such discoveries contend for testing of Remdesivir prior in COVID-19 [17]. Remdesivir didn't bring about huge decreases in SARS-CoV-2 RNA burdens or perceptibility in upper respiratory tract or sputum examples in this investigation in spite of indicating solid antiviral impacts in preclinical models of disease with coronaviruses. In African green monkey kidney Vero E6 cells, Remdesivir hindered SARSCoV- 2 with a half viable fixation (EC50) of 0.46 μg/mL and an EC90 of 1.06 μg/mL. In human nasal and bronchial aviation route epithelial cells, a fixed 20 μM (12.1 μg/mL) focus diminished assessed intracellular viral titres over 7.0 log10 half tissue culture infective portion per ml at 48 h.18 in human aviation route epithelial cells, the EC50 for Remdesivir was 0.042 μg/mL for SARS-CoV and 0.045 μg/mL for MERS-CoV. In a murine model of MERS, subcutaneous Remdesivir demonstrated critical antiviral and clinical impacts with a portion routine that kept up plasma fixations more prominent than 1 μM (0.60 μg/mL) all through the dosing interval. In rhesus macaques, a 5 mg/kg portion, answered to be generally proportionate to 100-mg day by day dosing in people, was viable for treatment of MERS-CoV contamination and decreased aspiratory infection replication when begun at 12 h after infection.18 Healthy grown-up volunteers getting dosages like our preliminary (200 mg on day 1, 100 mg on days 2-4) had mean pinnacle plasma convergences of 5.4 μg/mL (rate coefficient of variety 20.3) on day 1 and 2.6 μg/mL (12.7) on day 5.24 Doses of 150 mg/day for 14 days have been sufficiently endured in sound grown-ups, and a day by day portion routine of 150 mg for 3 days followed by 225 mg for 11 days had all the earmarks of being commonly very much endured in one patient with Ebola meningoencephalitis. However, the pharmacokinetics of Remdesivir in seriously sick patients, and especially the centralizations of the dynamic nucleotide metabolite (GS-441524) triphosphate in respiratory tract cells of rewarded patients, is obscure. Investigations of higher-portion regimens for which there are security information (eg, 150–200 mg day by day dosages) warrant thought in serious COVID-19. Our investigation found that Remdesivir was satisfactorily endured and no new wellbeing concerns were recognized. The general extent of patients with genuine unfavourable occasions would in general be lower in Remdesivir beneficiaries than fake treatment beneficiaries. Notwithstanding, a higher extent of Remdesivir beneficiaries than fake treatment beneficiaries had dosing rashly halted by the specialists in light of unfriendly occasions including gastrointestinal manifestations (anorexia, queasiness, and regurgitating), aminotransferase or bilirubin increments, and declined cardiopulmonary status. Restrictions of our investigation remember inadequate capacity to distinguish accepted contrasts for clinical results, commencement of treatment very late in COVID-19, and the nonappearance of information on irresistible infection recuperation or on conceivable rise of decreased vulnerability to Remdesivir. Of note, in non-human primates, the inhibitory impacts of Remdesivir on irresistible SARS-CoV-2 recuperation in bronchoalveolar lavages were a lot more prominent than in controls, however popular RNA recognition in upper and lower respiratory tract examples were not reliably diminished versus controls. Coronaviruses incompletely impervious to restraint by Remdesivir (around six-times expanded EC50) have been gotten after sequential in vitro entry, yet these infections stay helpless to higher Remdesivir fixations and show impeded fitness. The regular utilization of corticosteroids in our patient gathering may have advanced viral replication, as saw in SARS and MERS despite the fact that these investigations just revealed prolongation of the discovery of viral RNA, not irresistible infection [18]. Besides, we have no response to whether longer treatment course and higher portion of Remdesivir would be valuable in patients with serious COVID-19. In rundown, we found that this portion routine of intravenous Remdesivir was sufficiently endured yet didn't give critical clinical or antiviral impacts in genuinely sick patients with COVID-19. Be that as it may, we were unable to prohibit clinically important contrasts and saw numerical decreases in some clinical boundaries. Progressing concentrates with bigger example sizes will keep on advising our comprehension regarding the impact of Remdesivir on COVID-19. Moreover, methodologies to upgrade the antiviral intensity of Remdesivir (eg, higher-portion regimens mix with different antivirals, or SARS-CoV-2 killing antibodies) and to relieve immunopathological have reactions adding to COVID-19 seriousness (eg, inhibitors of IL-6, IL-1, or TNFα) require thorough investigation in patients with extreme COVID-19 (Figure 2) [19-26].

Figure 2: (a-d) Remdesivir and different drug actions on COVID-19.
While the ongoing examination on the empathetic utilization of Remdesivir for COVID-19 patients has demonstrated a 68% clinical improvement it is a one-arm study that renders the assessment of the viability in decreasing passing and the length of remain of hospitalization recalcitrant because of a lacking of the benchmark group. We concocted a two-arm controlled investigation configuration to reenact the rewarded and the untreated (control gathering) bunch by applying two individual progress models to the exact information on elements of the sickness seriousness) that are arranged into low-(no and low oxygen supplement), medium-(non-obtrusive ventilator and high oxygen supplement), and high-(ECMO and intrusive ventilator) from enrolment until release, demise or the finish of development. By utilizing a mimicked two-arm controlled investigation, the Remdesivir treatment bunch rather than the benchmark group prompted a factually fundamentally 29% (95% CI: 22-35%) decrease of death from COVID-19. The rewarded bunch likewise uncovered a 33% (95% CI 28-38%) fundamentally higher chances of release than the benchmark group. The middle opportunity to release for the rewarded gathering (5.5 days, 16.5 days, and 29.5 days for low-, medium-, and high-chance state, individually) was around half of those of the control arm. Our outcomes with a reenacted two-arm controlled examination have supported the viability of Remdesivir as well as made incredible commitment to planning a further enormous scope randomized controlled preliminary. They have noteworthy ramifications for lessening transmission likelihood and irresistible time of COVID-19 patients while reaching with powerless social insurance laborers during hospitalization.
Citation: Tiwari S, Sharma S (2021) Remdesivir Effectiveness against COVID-19. Gen Med (Los Angeles). 9: 352.
Received: 09-Jul-2021 Published: 30-Jul-2021, DOI: 10.35248/2327-5146.21.9.352
Copyright: © 2021 Tiwari S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.